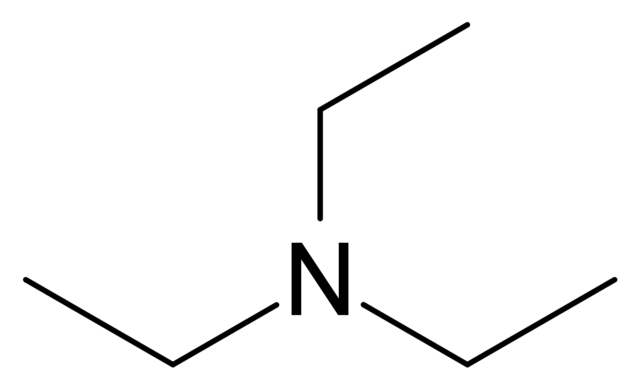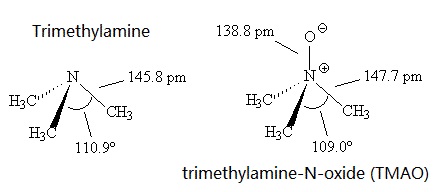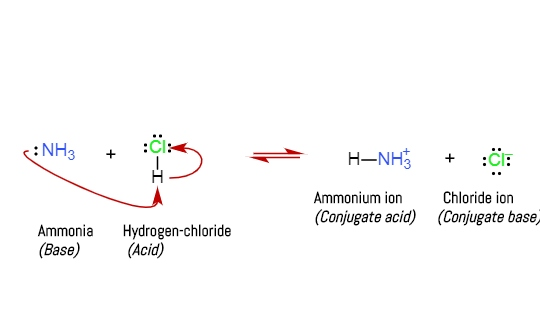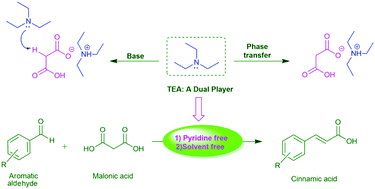
Triethylamine: a potential N-base surrogate for pyridine in Knoevenagel condensation of aromatic aldehydes and malonic acid - New Journal of Chemistry (RSC Publishing)

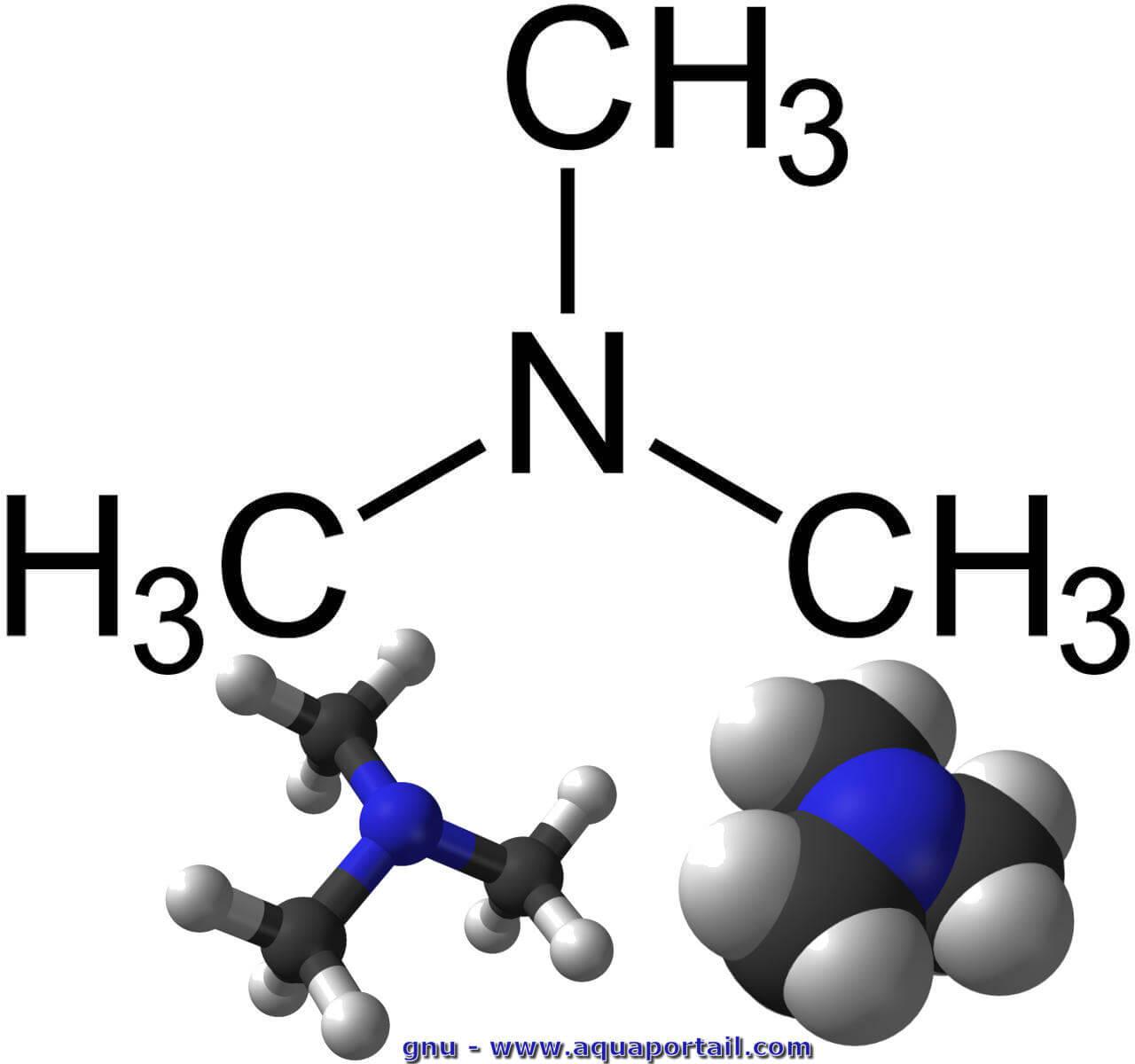
Trimethylamine Is A Nitrogenous Base And Can Be Protonated To Trimethylammonium Cation. This Colorless, Hygroscopic, And Flammable Tertiary Amine Has A Strong Fishy Or An Ammonia-like Odor Stock Photo, Picture And Royalty

Calculate the pH of a 0.443 M aqueous solution of trimethylamine. (Kb = 6.3 x 10-5) | Homework.Study.com

SOLVED: 'The compound trimethylamine, (CH3)3N, is a weak base when dissolved in water Write the Kb expression for the weak base equilibrium that occurs in an aqueous solution of trimethylamine: Kb'
✓ Solved: Base Ionization Trimethylamine, (CH3)3N, is a gas with a fishy, ammonialike odor. An aqueous...

mass spectrum of N,N-dimethylmethanamine (trimethylamine) C3H9N (CH3)3N fragmentation pattern of m/z m/e

What is the Difference Between Dimethylamine and Trimethylamine | Compare the Difference Between Similar Terms
Trimethylamine is a nitrogenous base and can be protonated to trimethylammonium cation. This colorless, hygroscopic, and flammable tertiary amine has Stock Vector Image & Art - Alamy
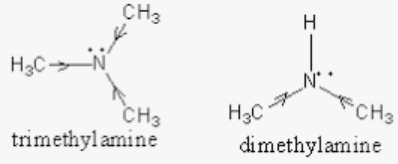
Which among the following is strongest base in gas phase? (a) Triethylamine (b) Diethylamine (c) Ethylamine (d) Ammonia