UV-Visible Spectrophotometric Method Development and Validation of Assay of Paracetamol Tablet Formulation | OMICS International
View of HPLC ASSAY OF MODEL TABLET FORMULATIONS CONTAINING METRONIDAZOLE AND CIPROFLOXACIN | International Journal of Pharmacy and Pharmaceutical Sciences

Are HPLC-UV Methods Fit for Purpose as True Arbiters of Quality for APIs? | American Pharmaceutical Review - The Review of American Pharmaceutical Business & Technology


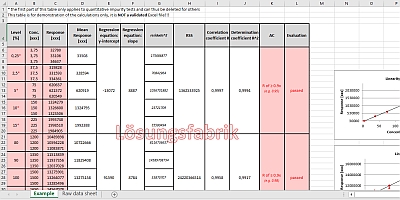




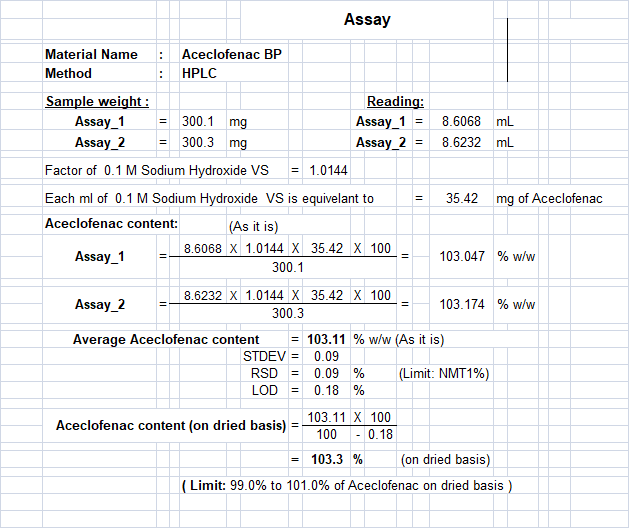
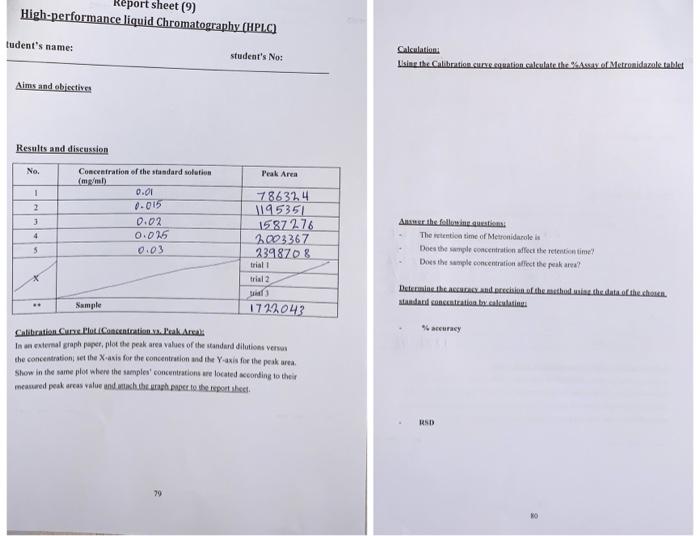


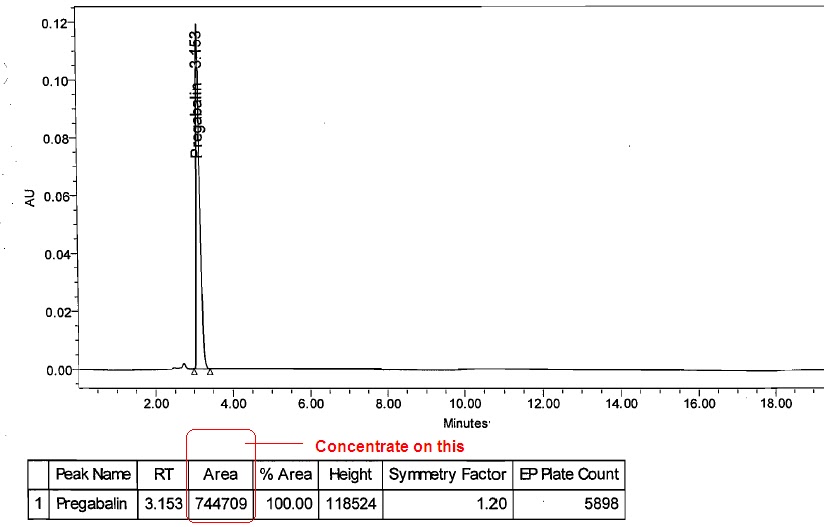
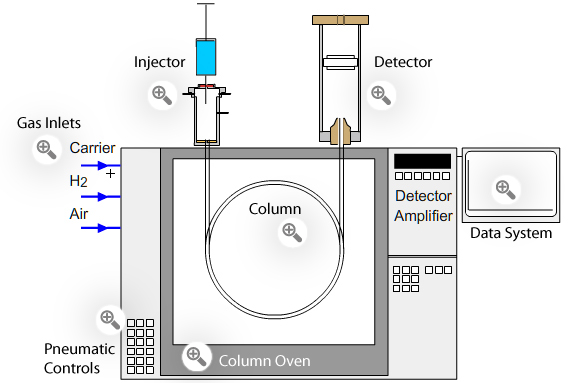
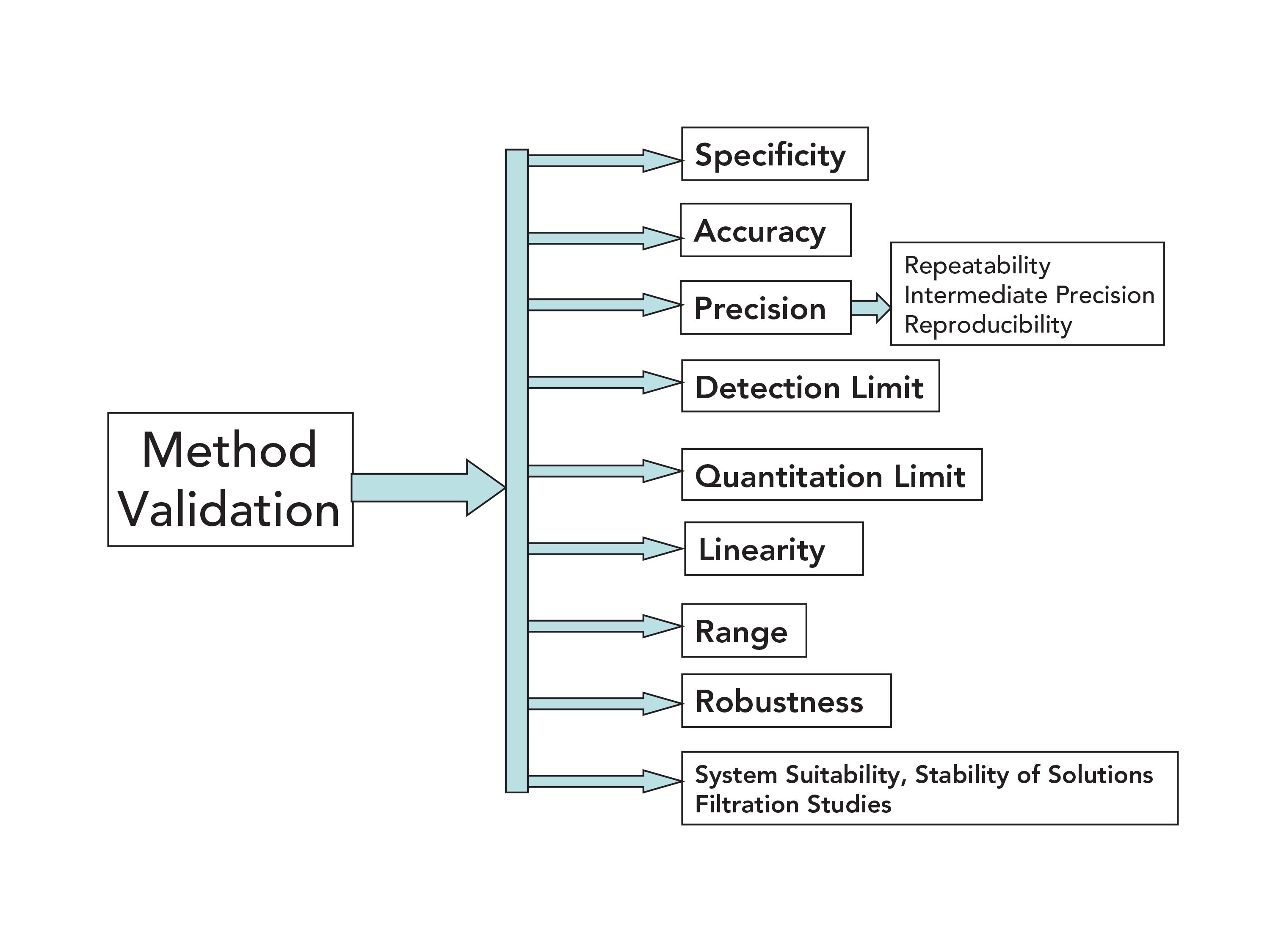



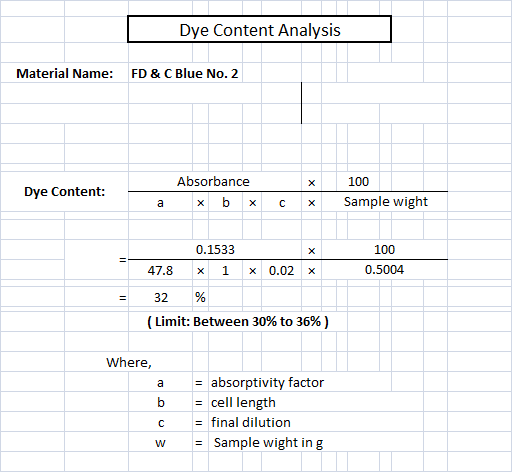
![LAB] Assay of Ascorbic acid with Calculations – Pharmawiki.in LAB] Assay of Ascorbic acid with Calculations – Pharmawiki.in](https://pharmawiki.in/wp-content/uploads/2014/01/Assay-of-Ascorbic-Acid-with-Calculations.jpg)