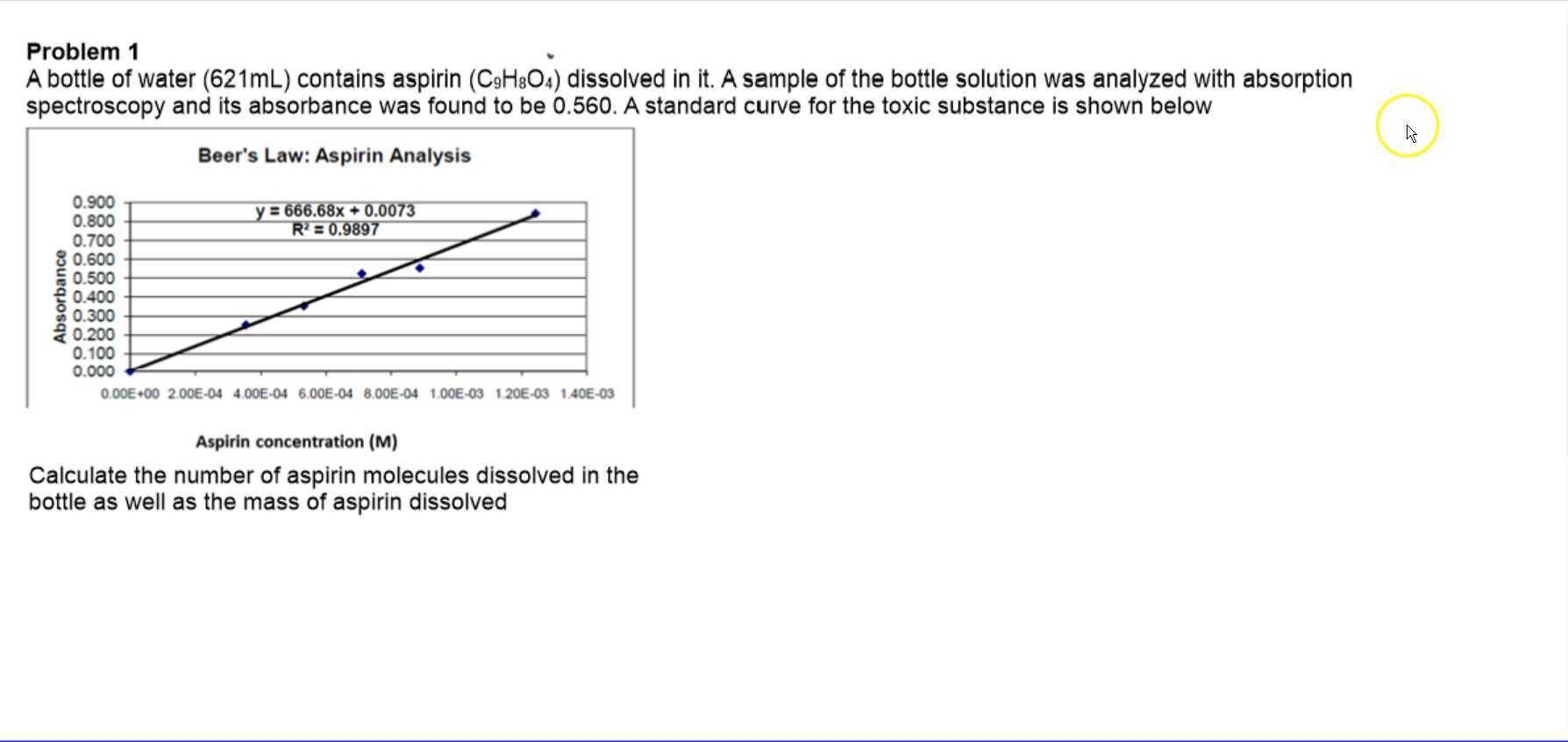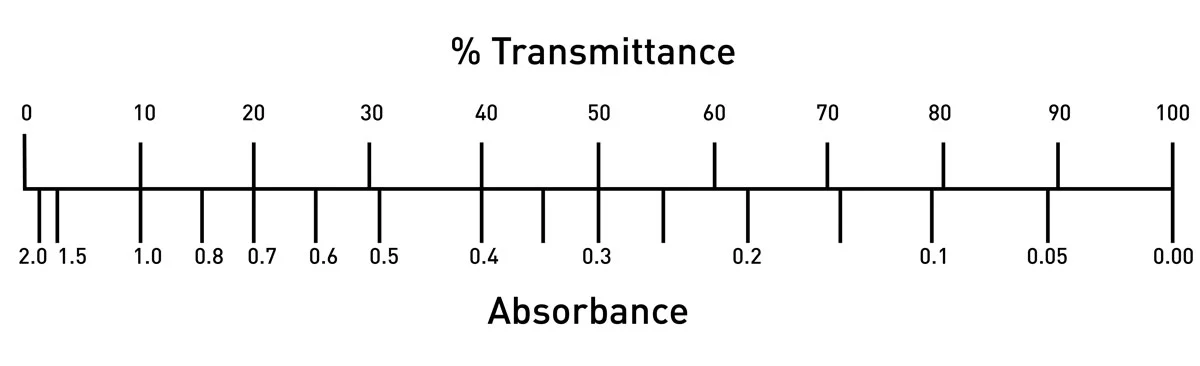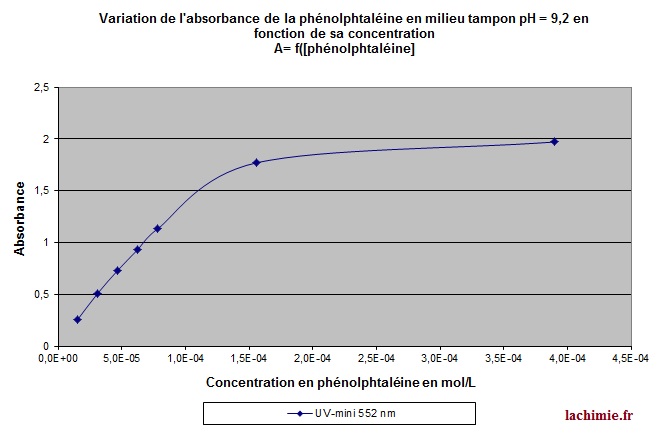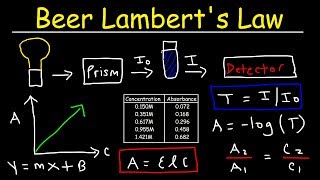
Beer Lambert's Law, Absorbance & Transmittance - Spectrophotometry, Basic Introduction - Chemistry - YouTube
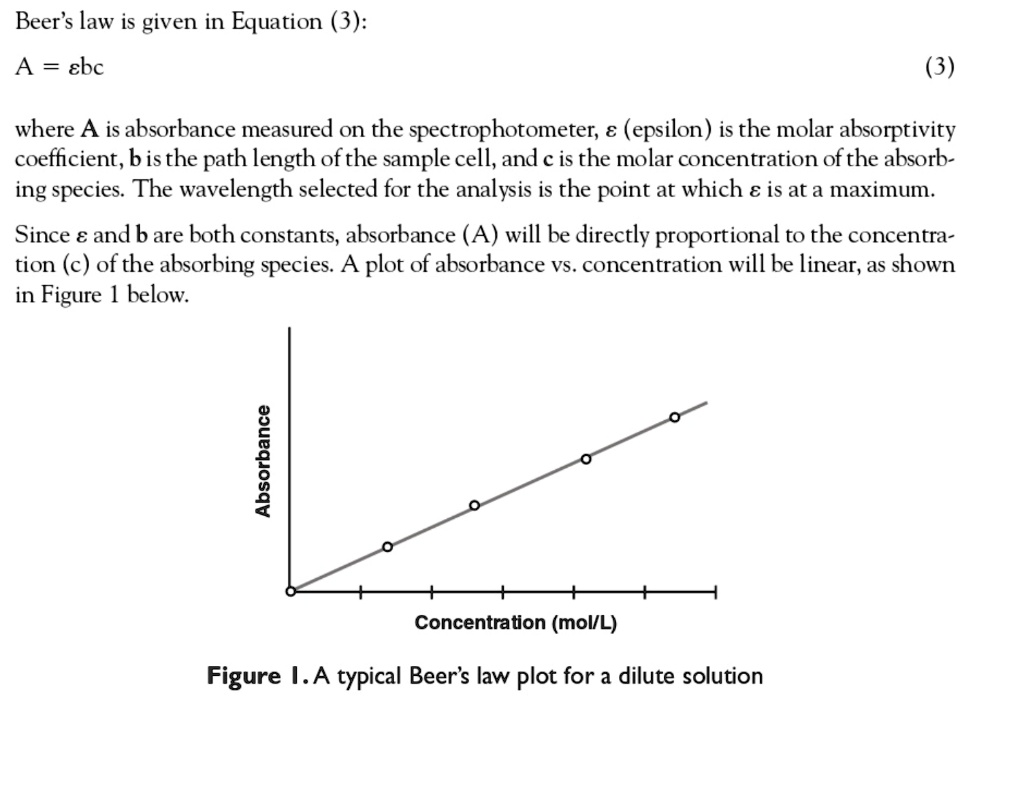
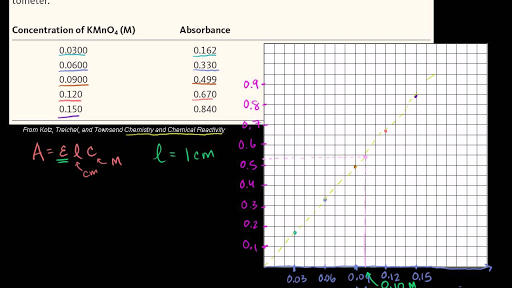
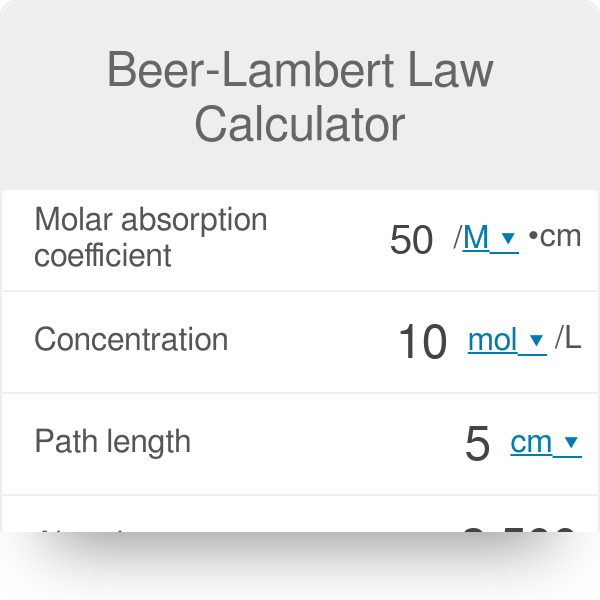
SOLVED: Beer's law is given in Equation (3): A ebc where A is absorbance measured on the spectrophotometer; (epsilon) is the molar absorptivity coefficient, bis the path length ofthe sample cell; and

The absorbance of the solution with epsilon value was calculated using... | Download Scientific Diagram

SOLVED: Absorption Max Wavelength (nm) 309 IQQUM Absorbance 2.29 5QuM Absorbance 1.13 25UM Absorbance 0.54 Unknown Absorbance 1.71 Slope 23.314 y-intercept Unknown EDMAB Concentration 0.0733 Using your value for molar absorptivity at
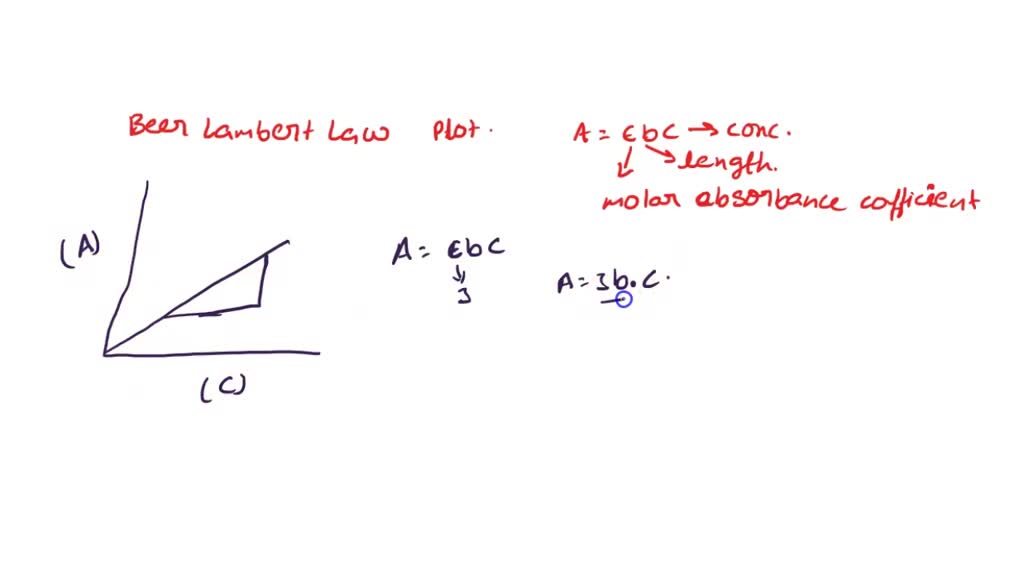
SOLVED: In the Beer-Lambert law plot, what does the x-axis represent from the absorbance equation (a=3bc)? What does the y-axis represent? In the equation for the straight line for the Beer-Lampert plot (